One of the rarely discussed issues with syringomyelia and Chiari malformations is that, in addition to (and because of) neural compression and hydrodynamic alterations, these diseases cause brain and spinal cord damage or injury. Treating the offending problem rarely reverses such injury. So, if the aim is to improve function in individuals affected by Chiari and syringomyelia, it is not enough to treat the abnormality itself. It is important that the resultant injury to the spinal cord, brainstem, or brain, heals as well. Unfortunately, unlike peripheral neurons, brain and spinal cord neurons are very resistant to repair. A large area of research has focused on experiments designed to optimize healing of the injured Central Nervous System (CNS) tissues. Some of the important issues related to this research are summarized here.
Over a century ago, a Spanish scientist, Ramón Y Cajal, was awarded the Nobel Prize in medicine or physiology for showing that, whereas injured peripheral nerves heal well after an injury, the adult brain and spinal cord show little or no recovery (6). To date, there is no cure for spinal cord injury (SC1) or stroke. In the US, there are about 4.4 million people with persistent disabilities due to stroke (3). Another 300,000 are living with various forms of SCI (7). Over the years, Cajal and many others have elucidated the myriad barriers to CNS repair. These have ranged from inhibitory molecules in the white matter or myelin (11), the physical barrier to growing axons called the glial scar that is created by an injury (5,12), and the intrinsic reduced potential for adult neurons to grow. It is thought that certain molecular switches are turned off after the CNS is formed, while the baby is in the womb, so that after birth and into adulthood the potential for neuronal repair declines. Interestingly, other animals like frogs and fish have been noted to retain the ability to regenerate and repair their brain and spinal cord throughout life. Within seconds to minutes of a stroke, traumatic injury, or other disorders of the brain or spinal cord, some neurons are permanently injured and die, while others are injured temporarily, and their fate depends on the treatment instituted, and how fast it is given (Fig.1). In instances of progressive spinal cord compression, such as syringomyelia, therapies tend to be directed at protecting surviving neurons within the vicinity of the injury (neuroprotection), the repair and elongation of neuronal extensions called axons (regeneration), or the formation of new neurons (neurogenesis)

It is crucial for therapies to be administered within hours of an acute injury, and as soon as possible in chronic injuries. Treatment in such cases may be aimed at reducing tissue swelling and dilating blood vessels to optimize glucose and oxygen supply. Moderating the invasion of dying tissue by inflammatory cells has also shown benefit. Importantly, the events implicated in tissue swelling and toxicity involve disordered cell membrane pumps, which regulate sodium, potassium and calcium physiology (4). This disables the cell membrane’s ability to control what goes in and out, hence the swelling and death. Several interventions, therefore, seek to correct this abnormal physiology by targeting pump function. The use of steroids to reduce swelling of the spinal cord following traumatic injury remains controversial.
As stated, neurons grow readily in the developing mammalian brain and spinal cord. This type of growth stops around birth and cannot be elicited again after an injury. Efforts in regeneration neuroscience seek to potentiate adult neurons to turn on neonatal pro-regenerative switches that lead to axonal elongation. Therapeutic interventions in this category have included growth factors like neurotrophins (2), folic acid (8), inflammation (9), over expression of vitamin A receptors (13), and conditioning lesions of peripheral nerves to stimulate regeneration within the CNS (10). Once neuronal growth is initiated, the axons need to grow to their rightful targets (in the right direction). As such, guidance cues called chemo-attractants have been studied as well (2)
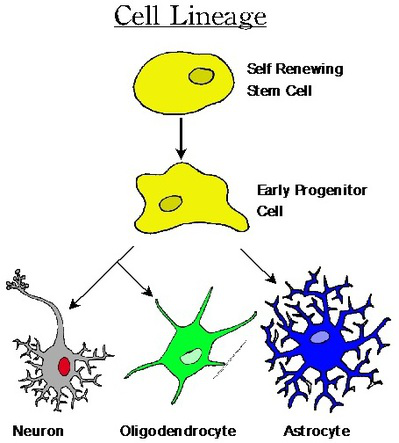
Contrary to what we thought over 20 years ago, new neurons continue to appear within specific regions of the brain and spinal cord throughout life (1). These neurons are derived from a form of stem cells, neural stem cells, which are found along the lining of the fluid cavities in the brain and spinal cord, and the hippocampus. The stem cells are also capable of forming astrocytes and oligodendrocytes, which are the supporting cells of the CNS (Fig.2). This is the basis for stem cell-based therapies. These range from embryonic stem cells, to those stem cells that reside in our CNS and can be activated later in life. Additionally, stem cell reprogramming in which skin cells can be modified to form stem cells is a rapidly evolving exciting new field. The future of this frontier, however, is quite complex. Before stem cells can become truly useful in treating medical conditions, the biology of the cells has to be better understood and controlled. Research goals include increasing and controlling the growth of stem cells, improving their survival, ensuring their conversion to neurons, and guiding their migration to and integration within the complex circuitry of the CNS (14).
With several of the aforementioned interventions, functional recovery has been observed in the hindlimbs and forelimbs of animals, with positive effects on walking, skilled movements, sensation, and other outcome measures. Indeed, experimental SCI is no longer incurable. The challenge is to optimize and understand these interventions, and translate some of this success to human applications. One of the most important lessons that researchers have learned along the way is that no single drug or intervention will lead to the cure of paralysis or stroke. Instead, a combination of therapies will be required. These include: 1) interventions that modify the neuronal environment to make it permissive for regeneration (neuroprotection, reduction of the glial scar, and elimination of the inhibitory factors in the myelin) and 2) interventions that potentiate adult neurons to regenerate. Scientists hope that, by understanding more about the life and death of neurons, they can develop new treatments, and possibly even cures, for brain and spinal cord diseases that affect the lives of millions of Americans and people around the world.